Design of Experiments (DoE), introduced in the 1920s, is a valuable modelling tool to support in amongst the systematic development of (bio)pharmaceutical products, processes and analytical methods. Especially in combination with risks assessments (e.g. FMEA), DoE is paramount of importance to establish quantitative relationships between potential critical parameters (related to e.g. product, process, method) and a defined response (e.g. product quality).
Compared to other industries, the (bio)pharmaceutical industry has been a relatively late adopter of DoE. Nowadays, DoE has become a recognized tool likely boosted by the implementation of Quality by Design (QbD), the approach to design a quality product and a well understood, controlled and high performing manufacturing process.
In the experience of Venn life Sciences scientists in Big pharma are well aware of the value of DoE in both early and late stages of development. DoE can support formulation development and serves to optimize and establish an optimal and robust set of operating conditions for manufacturing processes and analytical test methods. Scientists in Big pharma are often in the privileged position to have support from a statistical department which can help in the design of the experiments and result evaluation supported by advanced (costly) software, thereby lowering to barrier to use DoE.
For small and midsize (bio)pharma companies as well as CMOs, there is often some hesitation to use DoE. Various reasons for that may be given. One is that scientists are lacking the appropriate expertise to use DoE, often in combination with not having access to software programs to create designs and perform statistical evaluations. Stat-Ease, Minitab or JMP are examples of user-friendly software programs offering the most common experimental designs. Another reason for the slower adoption of DoE lies in the fact that smaller (bio)pharma companies and CMOs are often struggling when to best adopt DoE, either in early product -, process- or test method development or in later stage development. In our experience we often see that companies in early development perform too many (costly) experiments, often not in a very well-structured manner. In addition, in many cases the traditional one-factor-at-a-time (OFAT) approach is used which does not provide insight in potential interaction between factors. As a possible consequence follow-up studies may not confirm earlier experiments, or a process or analytical method is not as robust as anticipated.
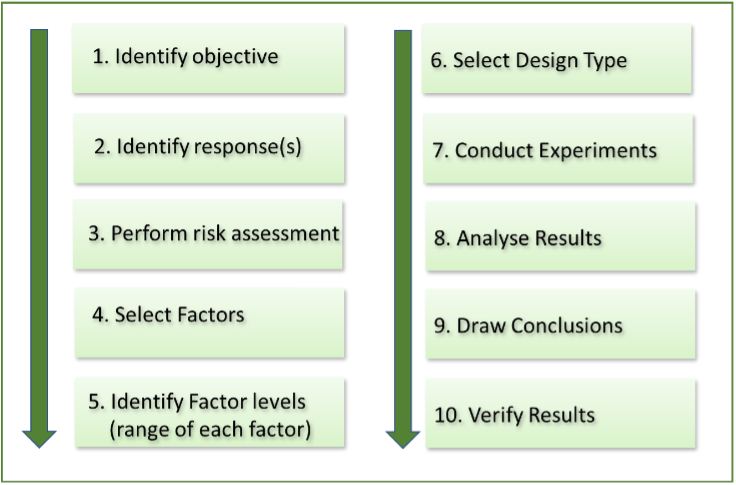
The application of DoE is highly beneficial for companies working at early and late stage (bio)pharmaceutical product, process and method development. The adoption of DoE will short track your development, saving precious resources and costs. Give it a try!

Gerben Wynia (MSc) is senior CMC consultant at Venn Life Sciences. He has over 25 years of experience in pharmaceutical R&D and has worked on the development of both small molecules and biomolecules in early and late stages of product development. He has expertise in the use of statistical methodology in the development of products, manufacturing processes and analytical methods.
If you are interested to learn more about how DoE can support your development activities, please contact Venn life Sciences at getintouch@vennlife.com. We would welcome the opportunity to discuss how DoE might be of benefit to your company’s development. Venn life sciences offers help in risk assessments, the set-up of DoE experiments, and statistical evaluation and interpretation of results.